-
510(k) SUMMARY OF SAFETY AND EFFECTIVENESS CozmoTM Insulin Pump (Model 1700) and Accessories I. GENERAL INFORMAT
ID: MzEgAiyaCv
From:
-
 Flash Glucose Monitoring: A Patient's and Clinician's Concerns Endocrine
Flash Glucose Monitoring: A Patient's and Clinician's Concerns Endocrine
ID: avyE7S8CQA
From:
-
 The Food and Drug Administration's (FDA's) 510(k) Process: A Systematic Review 1000 Cases - The American Journal of Medicine
The Food and Drug Administration's (FDA's) 510(k) Process: A Systematic Review 1000 Cases - The American Journal of Medicine
ID: LxB8hJMjgE
From:
-
Surveillance of Medical Devices: Comparison of Strategies in the EU, Japan, and China | PLOS Medicine
ID: UKpQQbQR2F
From:
-
 100 years of celebrating the past, present and future of diabetes | Medicine
100 years of celebrating the past, present and future of diabetes | Medicine
ID: PyjzFFpNZz
From:
-
ID: XEfRzldLvu
From:
-
REVIEW MEMORANDUM
ID: FcI3ye5N9C
From:
-
 Safety and efficacy of zilucoplan in patients generalised myasthenia gravis a randomised, double-blind, placebo-controlled, phase 3 study - The Lancet
Safety and efficacy of zilucoplan in patients generalised myasthenia gravis a randomised, double-blind, placebo-controlled, phase 3 study - The Lancet
ID: LoydP7UleG
From:
-
U §
ID: gEhKrv84zi
From:
-
 American Association Clinical Endocrinology Clinical Practice Guideline: The Use Advanced Technology in the Management of Persons With Diabetes Mellitus - Endocrine Practice
American Association Clinical Endocrinology Clinical Practice Guideline: The Use Advanced Technology in the Management of Persons With Diabetes Mellitus - Endocrine Practice
ID: 40QwhYzy8C
From:
-
 An FDA Viewpoint on Unique Considerations for Medical-Device Clinical Trials |
An FDA Viewpoint on Unique Considerations for Medical-Device Clinical Trials |
ID: QLXqHY4NNU
From:
-
Guidance for
ID: xL6tOB0ksV
From:
-
 Management diabetes and hyperglycaemia in the hospital - The & Endocrinology
Management diabetes and hyperglycaemia in the hospital - The & Endocrinology
ID: XJ4Su3ZIcx
From:
-
510(k) Summary of and Effectiveness
ID: z3bedhQtYJ
From:
-
 Design and analysis of high-risk device clinical trials for diabetes monitoring and treatment: a review | Journal Pancreatology
Design and analysis of high-risk device clinical trials for diabetes monitoring and treatment: a review | Journal Pancreatology
ID: Ox8sOj0KlD
From:
-
 Template Version 1.0 - Accessdata Fda - Fill and Sign Printable Template
Template Version 1.0 - Accessdata Fda - Fill and Sign Printable Template
ID: 1pqNb03Aq5
From:
-
 An FDA Viewpoint on Unique Considerations for Medical-Device Clinical Trials |
An FDA Viewpoint on Unique Considerations for Medical-Device Clinical Trials |
ID: xqDcUSCLlV
From:
-
Artificial Device Systems
ID: Oolyj0TQjx
From:
-
SUMMARY OF EFFECTIVENESS (SSED)
ID: c4cTFn6gob
From:
-
Diabetes Tests, Programs Supplies of 44 07/06/2015 11:29 AM
ID: g4ygLB4zxJ
From:
-
 FDA Regulation of Medical Devices -
FDA Regulation of Medical Devices -
ID: azfeSY5CM5
From:
-
 July 2011 digital edition of Practice by McMahon Group - Issuu
July 2011 digital edition of Practice by McMahon Group - Issuu
ID: wP9YcSMwq2
From:
-
Hearing Aids and Wearable, Bone Anchored and Semi-Implantable – Exchange Medical Policy
ID: wRSICCRqlL
From:
-
 How FDA Regulates Intelligence in Medical The Pew Charitable Trusts
How FDA Regulates Intelligence in Medical The Pew Charitable Trusts
ID: 1Jvb3iuKI5
From:
-
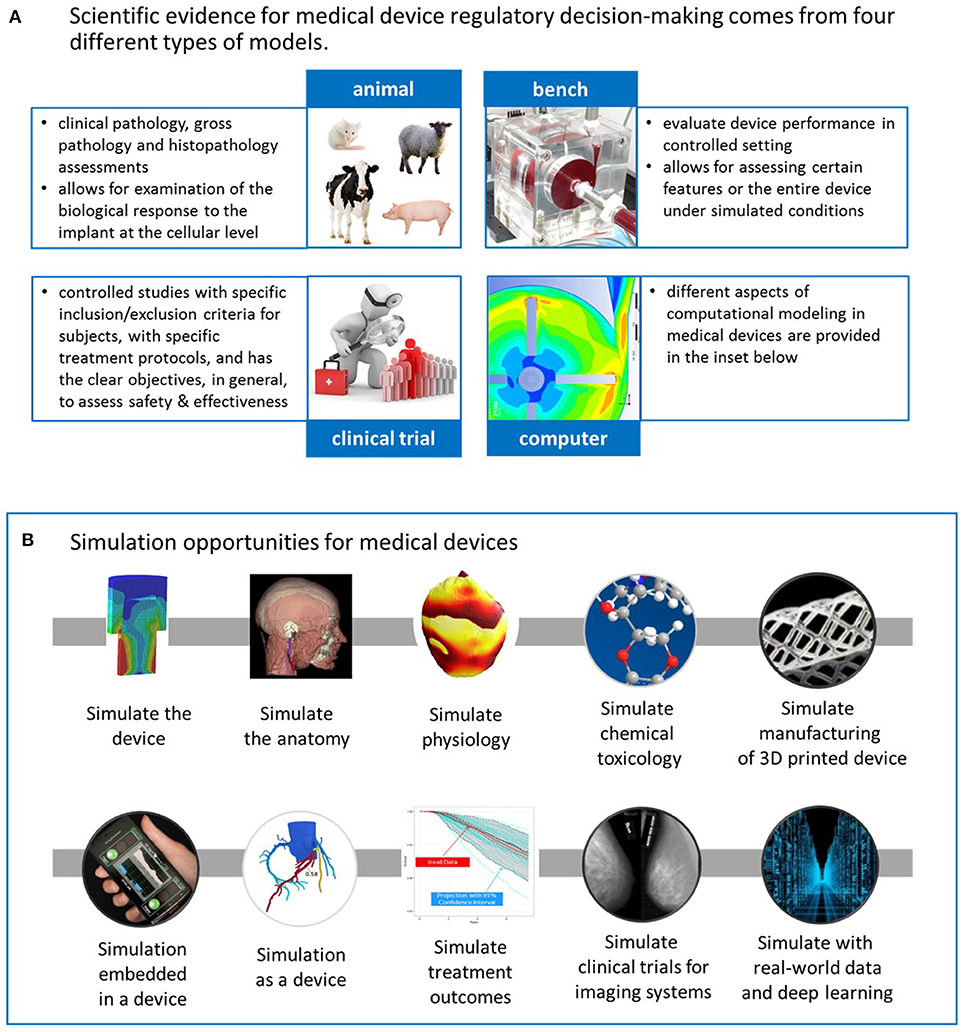 Frontiers | Advancing Science With Computational Modeling for Medical Devices at the FDA's Office of Science and Engineering Laboratories
Frontiers | Advancing Science With Computational Modeling for Medical Devices at the FDA's Office of Science and Engineering Laboratories
ID: tzWpjg1sBG
From:
-
 Study Design and Data Analysis of Artificial Device Systems with Closed-Loop Glucose-Sensing Delivery
Study Design and Data Analysis of Artificial Device Systems with Closed-Loop Glucose-Sensing Delivery
ID: szp83Fjt0H
From:
-
 GORE® TAG® Thoracic Stent with ACTIVE CONTROL System | Medical
GORE® TAG® Thoracic Stent with ACTIVE CONTROL System | Medical
ID: zAL1VLkcNq
From: